John A. Medeiros
![]()
Color Deficit Vision
No discussion of color vision models can be complete without an accounting of how color vision function can go wrong and “color blindness” results. There are a number of ways in which color vision function fails, but by far the most common are the two types of red-green deficit vision, protanopia and deuteranopia. Both of these forms exhibit a gradation from nearly normal color vision through their anomalous variations (protanomolous or deuteranomolous) to the near complete loss of the ability to distinguish reds and greens. These forms have X-linked recessive genetics and affect around 8% of the male population and a much smaller percentage of females.
Color deficit vision is by itself a major topic of investigation with a vast body of research and published literature. I can hardly hope to do justice to all of that work in this short monograph. However, I will address a couple of topics about color blindness in terms of the CSM model, most of which is covered in more detail in the book, Cone Shape & Color Vision.
Within the CSM model, a direct way in which the cones will fail to provide the necessary time-ordered color information for the retinal neural circuitry to interpret appropriately is for the cones to be the wrong size to optimally disperse the spectrum along their length. This could happen in either of two ways; they could be too small or they could be too large. Either would cause deficient color vision in subtly different ways. It should be noted that this incorrect sizing might have nothing to do with the cone’s physical size but be caused by incorrect values of the refractive index difference between the cones and their surrounding interstitial medium. Recall that the index of receptor “size” is the dimensionless waveguide parameter V,
V= (πd/λ)(n12– n22)1/2.
For a given cone diameter, d, at a particular location along its length and for cone refractive index n1, then if the surrounding medium refractive index, n2, is larger (smaller) than normal, the cones will be smaller (larger) than they should be for optimum operation.
If the cones are too small, than long wavelength (red) light will be very inefficiently coupled into the cone’s photosensitive outer segment. This will have two immediately obvious consequences: a lower sensitivity to red light and inefficient use of the cone length for discrimination. Both of these characteristics are signatures of one type of color deficit vision, protanopia, where there is both low sensitivity to red light and poor color discrimination.
If the cones are too large, then all colors will couple efficiently to the cone’s photosensitive outer segment, but the spectrum will be dispersed (at least in the lowest-order, HE11 mode) inefficiently over the cone with mode cut off dispersing the spectrum only over the lower portions of the cone. The immediately obvious consequences of this mistuning will be reduced color discrimination and no reduction in spectral sensitivity, both of which are signature characteristics of deuteranopia.
An idea of how this cone mistuning might look is provide by the accompanying figure. Here, I have taken the photograph of spectral dispersion in a tapered fiber (shown at the beginning of this document) and simply cut a section from the photo and pasted it three different times into the figure. The portion pasted in the middle is meant to represent what the span of spectral dispersion due to mode cutoff would look like for optimum operation in the normal cones. The cone on the left is a portion cut out further down the fiber showing the spectral dispersion span if the cone is too small for optimum operation as in protanopia. On the right is the representation for the cone being too large as in deuteranopia. Note that in the too-small case, red light is not efficiently coupled into the cone and its spectral span is limited. In the too-large case, while the entire spectrum is coupled into the cone, the span is not ideally placed for optimum operation and there will be confusions due to second-order mode coupling, even for wavelengths that are not in the violet as in the normal cones.
In any case , in the context of the CSM model, if one can purposefully alter the refractive index of the cone’s surrou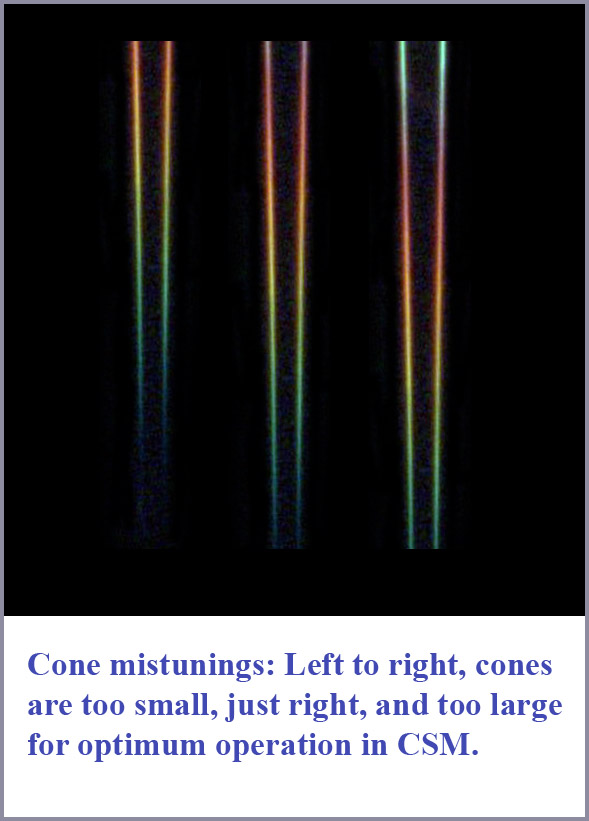 nding medium appropriately, it is not difficult to imagine that useful color vision function could be restored for “color blind” individuals. To “cure” protanopia one would need to increase the refractive index difference between the cones and its surround. This could be accomplished by increasing the refractive index of the interstitial medium by increasing the amount of included solids or suspended complex molecules in the medium. Conversely, to treat a deutan defect, it would be necessary to decrease that refractive index difference by reducing the refractive index of the surrounding medium by removing suspended solids or complex molecules.
nding medium appropriately, it is not difficult to imagine that useful color vision function could be restored for “color blind” individuals. To “cure” protanopia one would need to increase the refractive index difference between the cones and its surround. This could be accomplished by increasing the refractive index of the interstitial medium by increasing the amount of included solids or suspended complex molecules in the medium. Conversely, to treat a deutan defect, it would be necessary to decrease that refractive index difference by reducing the refractive index of the surrounding medium by removing suspended solids or complex molecules.
It may be of interest that this may actually have already been successfully demonstrated. In a number of papers published in the 1970’s, Louis F. Raymond reported on the treatment of patients with allergies who were also colorblind (Raymond, 1971, 1972, 1975). He cites one case (Raymond, 1975) of a patient who tested as red-green color deficit on both Ishihara plates and Hardy-Rand-Rittler Plates. The patient tested positive for allergies to bacterial endotoxins and air-borne pollens in intradermal tests. Hyposensitization treatment, consisting of administration of diluted antigenic solutions of the items to which he was allergic cured the colorblindness. He claimed that the patient’s color vision was normal on testing after two months, one year, and at two years later. He noted that he treated a total of 24 such cases with similar results.
There are a number of identifiable antibodies in human sera. Chemically, these are glycoproteins with molecular weights of around 150 kDa. These antibodies are a variety of immunoglobulins, including immunoglobulin E (IgE). It has been shown that IgE levels are reduced following hyposensitization treatments. Now the immunoglobulins are glycoproteins with oligosaccharide chains (sugars) covalently attached to their polypeptide. The heavy molecular weight molecules with attached sugars can be expected to have a similar effect on the refractive index of a medium as the acid mucopolysaccharides (MPS) that are known to be present in the interstitial medium of the retina. In addition, immuboglobulins have been directly observed to be present in the medium between the photoreceptors (Hollyfield, 1999). Conceivably then, the hyposensitization treatments may have altered (decreased) the refractive index of the interstitial medium and thereby restored color discrimination function through the reduction of high molecular weight proteins suspended in the sera and thus the medium around the photoreceptor outer segments.
If these results can be confirmed, and, if this is indeed a mechanism for restoration of color vision function, then the patients that would have benefited from this particular approach should be those with protanopia. That is, decreasing the density of the interstitial medium through the injections should decrease the refractive index of surrounding medium and thus increase the effective size of the cones. This could be expected to restore the subject’s color vision function so long as the appropriate retinal circuitry was still present. Unfortunately, Raymond never specified which type (or types) of the red-green color deficit vision he was able to treat. In any event, given the large number of individuals in the general population afflicted with color deficit vision, this is clearly an area of clinical research that should be further explored.
In this context, it is worth noting that the cones apparently maintain an elaborate machinery to regulate the refractive index of their surrounding medium. It has been demonstrated that the inner segments of the rods and cones are particularly rich in ribosomes and associated membranes that support the polymerization of activated monomers into high-molecular-weight MPS (Fine & Zimmerman (1963). In addition, the Golgi complex, which is generally considered to play an important role in secretory activities of a cell, is very prominent at the junction between the inner and outer segment. All of this strongly suggests that this is the site for secretion of the MPS that essentially controls the refractive index of the medium surrounding photoreceptor outer segments and thus the critical optical "size" of the cones.
While the physiological function of the MPS surrounding the photoreceptors has been described as something of a puzzle (Hall & Heller 1969), it would seem that in the context of the CSM model it would have the important role of regulating cone "size". Note that there is a class of genetically determined diseases in which there is an abnormal accumulation of MPS in various tissues – the mucopolysaccharidoses (Hurler’s syndrome). While the ocular symptoms in these syndromes are generally rather severe, it is of interest that the genetic transmission in most cases is X-linked recessive, just as in the common forms of red-green deficit vision. Perhaps the connection between color blindness and the machinery regulating the refractive index around the cones may indeed be more than superficial.
Another version of color deficit vision is true color blindness or achromatopsia. There have been a number of cases of complete color blindness where the subjects donated their eyes for scientific research after death. The autopsy results showed, in some cases, a normal population of cones, and in others a drastically reduced number of cones (Alpern et al, 1960; Falls, et al, 1965; Glickstein & Heath, 1975; and Harrison et al, 1960) . However, one common feature found by all the studies was that what cones were present were abnormal in shape with outer segments that were either grossly misshapen or abnormally squat and short. Whatever else may have been wrong in these eyes, it is quite clear that in the context of the CSM model such cones could not have provided useful color discrimination.